Aquaculture
Properly sited and managed, unfed aquaculture of organisms like seaweed and bivalves, as well as certain finfish, can complement wild seafoods and poses a minimal threat to ocean ecosystems. Carnivorous open net pen aquaculture, however, requires chemicals and fish-sourced feed, releases pollution, and risks fish escapes, as does “ranching” – the capture and captive fattening of wild fish. Not only do those practices threaten the ocean’s health, they also remove nutritious food from our system.
Smart regulations that foster ocean-friendly aquaculture, ban the most damaging practices, and mandate mitigation of environmental impacts are essential for healthy oceans and the people who rely on them. Specifically, responsible aquaculture policy:
- Promotes aquaculture systems that prioritize food and nutrition security by adding to the world’s food supply instead of taking from it;
- Protects ocean habitats by siting installations outside of sensitive areas and wildlife migration corridors, and mandates mitigation during site construction and operation;
- Avoids harming native wildlife through exposure to disease, escaped farmed species, entanglements, or farm waste
- Avoids the use of antibiotics, pesticides and harmful chemicals because of their risks to ocean and human health; and
- Does not displace small-scale fishers by privatizing coastal zones or create other, additional inequities in the name of “blue growth.”
WHAT IS AQUACULTURE?
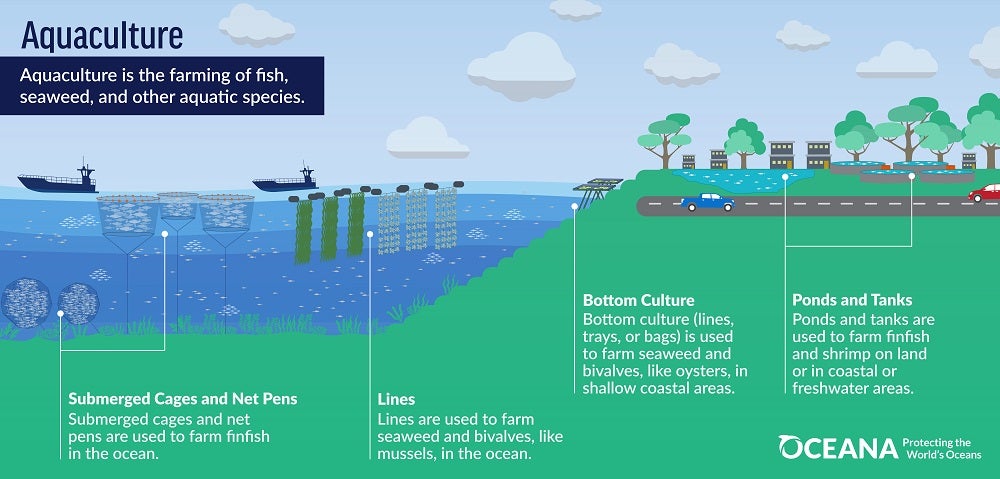
Aquaculture is the farming of fish, seaweed, and other aquatic species. This diverse and growing industry produced 130.9 million metric tons 1 (mt) of global fisheries and aquaculture produce in 2022 (FAO, 2024).
Over 500 aquatic species are farmed around the world – in the ocean, along the coast, or in freshwater on land. Of aquatic animals farmed for food, about 45% (59 million mt) – comes from inland ponds or tanks growing freshwater finfish like carp and tilapia. Marine and coastal aquaculture, sometimes called mariculture, makes up the remaining 55% (72 million mt) and includes seaweeds and microalgae (35 million mt), bivalve mollusks (18.7 million mt, including oysters and mussels), finfish (8.6 million mt, mostly salmon) and crustaceans (7.6 million mt, mainly shrimp) (FAO, 2024).
Almost three-fourths of animal aquaculture production (68 million mt) requires feed inputs, generally in the form of fresh, farm-made, or commercial pellet feeds, often including fishmeal and fish oil (FMFO) derived from wild fisheries. This “fed” aquaculture includes freshwater fishes like tilapia, catfish, and omnivorous carps, as well as carnivorous species that typically require higher FMFO inputs. Carnivorous aquaculture includes predatory finfish (like salmon, trout, grouper, and seabass), and certain shrimp, crabs, and prawns. Ranching – the practice of capturing juvenile predators, particularly tunas, and fattening them in offshore pens before being sent to market – also requires manufactured feed. Fed aquaculture is much more resource-intensive than unfed aquaculture but also tends to be more lucrative, driving its faster expansion (FAO, 2024). “Unfed” species, which filter particles from the water, make up the remaining third of farmed aquatic animals by weight (about 25 million mt). Bivalve mollusks like oysters and mussels comprise the bulk of unfed aquaculture, though it also includes filter-feeding freshwater finfish like silver and bighead carp. Seaweed aquaculture, including the farming of marine macroalgae like kelp, is also a form of unfed aquaculture, requiring little but space and sunlight. Farmed seaweeds add over 35 million metric tons to the world’s aquaculture harvest, about 30 times the amount of seaweed gathered from the wild. Because it requires so few inputs, unfed aquaculture is more likely to be environmentally friendly and low-impact, when sited and managed properly (FAO, 2024).
AQUACULTURE HAS A WIDE RANGE OF IMPACTS
The environmental footprint of aquaculture can vary 100-fold between products, depending on the species and the manner in which it is farmed (Hilborn et al., 2018). Some forms of aquaculture benefit the wider ecosystem when managed properly, while others can place a severe strain on local habitats, wildlife and natural resources. It is worth recognizing that some practices, by their very design, will never fully avoid or mitigate the impacts noted here no matter how careful.
Delivery of ecosystem services
Certain forms of aquaculture, if managed responsibly, can support healthy ecosystems by filtering water and sequestering carbon. Farming of filter-feeding bivalves, like mussels and oysters, can improve water quality by helping control excess nutrients. For example, oyster farming in the United States’ Chesapeake Bay helps restore the Bay’s depleted native oyster population and, in turn, the water filtration services those oysters provide (Parker and Bricker, 2020).
Seaweed farming can mitigate the acidification caused by the ocean’s absorption of anthropogenic carbon dioxide, taking up the gas during photosynthesis which increases the oxygen content and decreases the acidity of surrounding waters (Mongin et al., 2016). Seaweed can also be converted into biofuel, thus reducing net carbon emissions if used in lieu of fossil fuels (Duarte et al., 2017; Marquez et al., 2015), and even has the potential to replace resource-intensive fertilizers (Pechsiri et al., 2016).
Seaweed and bivalve aquaculture require no feed, no freshwater, and little fuel to produce (Williamson et al., 2015). With the right placement and management to protect sensitive habitats and wildlife, they can be a boon for the ocean.
Habitat loss
The establishment and operation of fish farms can destroy or degrade local habitats, not just through direct destruction, but via effluent runoff, siltation of nearby areas, or other knock-on effects. Aquaculture development is a major driver of mangrove habitat destruction. In the last 50 years, 20% to 35% of mangroves have been lost with over half of all mangrove destruction due to aquaculture (Goldburg et al. 2020). In tropical and subtropical areas, where farms are often unplanned and unregulated, 38% of global mangrove habitat loss is attributed to shrimp aquaculture development (McSherry et al. 2023, Ahmed et al., 2018).
Water pollution
Chemicals and waste products in aquaculture effluent can cause toxic algal blooms, oxygen-free dead zones in the water, and other severe environmental harm. Aquaculture farms use products like chlorine and copper, which can be toxic to aquatic life, for cleaning ponds, tanks, and equipment. Finfish and shrimp farms generate excess nutrients in the form of uneaten food pellets and animal waste, which can be flushed into nearby waters. This nutrient loading contributes to eutrophication – the rapid growth of algae and plants that then stifle and smother animal life. Among farmed fish, tilapia and carp aquaculture has especially high levels of nutrient runoff (Hilborn et al., 2018).
Land-based recirculating aquaculture systems can allow for containment and runoff prevention but are often energy intensive (Kim and Zhang. 2018).
Fish escapes & invasions
Unless wholly contained in land-based tanks, farmed fish will inevitably enter nearby ecosystems. Those fish can transmit disease to wild populations, compete with or displace local species, or interbreed with wild populations, affecting those populations’ viability.
Famously, the Nile perch’s introduction to Lake Victoria for fish farming led to the extinction of the native local cichlid fish. Even when escapees do not establish themselves as invaders, they pose problems: for instance, when non-native salmon escape their aquaculture pens in Chile, they threaten native species by preying on fish important to local fishers. In 2018, about 800,000 Atlantic salmon escaped in a single event in Chile, less than half of which were recovered (Quiñones et al., 2019). From 2011-2021, 1.73 million farmed salmon escaped from Norwegian salmon farms (Strand et al., 2024). Although they belong to the same species as their wild counterparts, their genetic differences mean that interbreeding threatens to erode the native population’s genetic structure (Taranger et al., 2015). In many countries, including Chile, Norway, and Canada, farmed fish are known vectors of disease to wild fish (Krkosek et al., 2024).
Disease, antibiotics & pesticides
Disease spreads rapidly among densely packed captive animals and can spread to wild fish (Barrett et al., 2019). While antibiotic and pesticide use can prevent and treat outbreaks, they also leach into surrounding environments. Salmon farms in Chile are one of the worst offenders: In 2015, they used 1,400 times more antibiotics than salmon farms in Norway. Per ton, Chilean salmon farms may use more antibiotics than any other meat producer. This intensive use of antibiotics breeds antibiotic-resistant bacteria and these “superbugs” have been found around salmon farms (Daoud et al., 2018; Shah et al., 2014), posing a direct risk to farm workers and raising concern about their further spread.
Pesticides can kill wild species in surrounding waters – the chemicals used to treat sea lice infestations also harm wild populations of crabs and other crustaceans (Bloodworth et al., 2019; Daoud et al., 2018). Parasites and diseases escape to surrounding waters and infect native species. In Norway, salmon lice have not only developed resistance to anti-louse pesticides but also spread to and infected wild salmon populations (Fjørtoft et al., 2019; Jensen et al., 2020).
Dangers to wildlife
Aquaculture farms create a risk of entanglement for marine wildlife and can pose a barrier to migrations (Clement and Sagar, 2013). The constant presence of fish attracts wildlife (Barrett et al., 2019)– even as farms need to deter those predators. As a result, fish-eating wildlife like seals, sea lions, dolphins, herons, and cormorants maybe killed inadvertently or illegally (Callier et al. 2017). Attempts to deter marine mammals with noise may constitute harassment and can alter feeding and other behaviors (Forney et al., 2017).
Mussel farms alone have fatally entangled an endangered North Pacific right whale in South Korea, an endangered leatherback turtle in Canada, a humpback whale in Iceland, and a Bryde’s whale in New Zealand (Young, 2015). These tragic events underline how essential proper siting and management is for even the “best” kinds of aquaculture.
Uses of wild-caught fish in aquaculture
Much of fed aquaculture relies on feed derived from wild-capture fish, which, if unsustainably managed, can lead to depletion of wild fisheries and the predators – including whales, seabirds, and commercially important fish – who rely on those forage species. In 2022, 11% of total global fish production (about 21 million mt) was used for non-food purposes, the majority of which (17 million mt) was reduced to FMFO. An estimated 74% of the world’s fish oil is used for aquaculture (FAO; 2024).
Despite technological advances that have reduced the proportion of FMFO in feed and decreased the overall food conversion rate (the ratio of food input to fish produced) from 3:1 to 1.3:1 (FAO, 2018), total aquafeed production still grew from 8 million mt in 1995 to 48 million mt in 2015 (FAO, 2018). Feed input requirements, particularly for carnivorous fish like salmon, still make for a net loss of fish protein, and increase pressure on wild fish stocks with 4kg of wild fish needed to produce 1 kg of farmed fish (Majluf et al., 2024).
In addition to the use of wild fish for animal feed, some types of aquaculture represent a further source of pressure on wild stocks because they rely directly on wild seed or larvae, or the capture and ranching of juvenile or adult fish that are then fattened in pens (Richardson et al., 2023).
Other resource demands
Aquaculture feeds also typically contain ingredients from terrestrial agriculture, such as soy, which bring their own environmental footprint. Recognizing this, some aquaculture feed suppliers are taking steps to reduce that harm, such as Brazilian soy suppliers to the salmon industry shifting away from growing soy on deforested land. Most aquaculture operations also require fuel or electricity (Hilborn et al., 2018), and land-based and freshwater farms can consume freshwater resources.
A range of socio-economic impacts
Socio-economic impacts of aquaculture run the gamut from grave injustice to sustainable jobs generator. When aquaculture is established in areas that otherwise support wild fisheries or tourism, its development can result in job losses for those sectors, even while providing jobs in its own right. Shrimp farm development in Asia and Central America can destroy mangroves and threaten the livelihoods of local and indigenous fishing communities who depend on them, and when communities protest, some have been met with abuse or murder. In the rush to create “blue economy” jobs through aquaculture, extant small-scale fishing communities may lose access to their fishing grounds (Belton et al., 2020).
In some cases, aquaculture is converting a high-quality, cheap, nutritious protein source, like sardines or anchovy, into one that is either lower quality and less nutritious, like tilapia, or a high-quality luxury item that few can afford, like farmed salmon. In Chile, the majority of aquaculture production is of salmon, which uses industrial methods in remote locations and is sold at a high price. Instead of producing fish for Chile and the region’s nutritionally-vulnerable population, farmed salmon is mainly exported for wealthy consumers abroad (Golden et al., 2017; Nahuelhual et al., 2019). Peru and Belize were also among the countries identified in a study of seafood-reliant, nutritionally-vulnerable nations as having high-value, export-oriented aquaculture with more than 50 percent of production by volume going to international markets (Golden et al., 2017). A similar pattern is unfolding in West Africa, where 40 industrial fish reduction plants have begun turning sardinella, a widely consumed local staple, into FMFO for export (Pauly, 2019), instead of prioritizing wild fisheries and smart aquaculture production as a potential solution for combatting the region’s food insecurity and malnutrition.
However, the conscientious development of nutrition-sensitive aquaculture may benefit communities by introducing job opportunities, new sources of income, and increased micronutrient intake, while reducing environmental impacts (Genschick et al., 2015; Shepon et al., 2020). The Philippines and China have large domestic-oriented aquaculture industries (focusing on low-value freshwater species) where aquaculture supports the needs of the nutritionally vulnerable (Golden et al., 2017). Seaweed farming is providing a more sustainable alternative livelihood for people, including former fishers, in places like Belize and the northeast United States.
Media Contact
Gillian Spolarich – gspolarich@oceana.org
Sources
Ahmed N, Thompson S and Glaser M (2018) Integrated mangrove-shrimp cultivation: Potential for blue carbon sequestration. Ambio 47: 441–452. doi: 10.1007/s13280-017-0946-2
Barrett LT, Swearer SE and Dempster T (2019) Impacts of marine and freshwater aquaculture on wildlife: a global meta‐analysis. Reviews in Aquaculture 11: 1022–1044. doi: 10.1111/raq.12277
Belton B, Little DC, Zhang W, et al. (2020) Farming fish in the sea will not nourish the world. Nature Communications 11: 5804. doi: 10.1038/s41467-020-19679-9
Bloodworth JW, Baptie MC, Preedy KF and Best J (2019) Negative effects of the sea lice therapeutant emamectin benzoate at low concentrations on benthic communities around Scottish fish farms. Science of The Total Environment 669: 91–102. doi: 10.1016/j.scitotenv.2019.02.430
Clement D and Sagar P (2013) Literature review of ecological effects of aquaculture: Effects on marine mammals. New Zealand: Ministry for Primary Industries.19. ISBN 978-0-478-38817-6 (online)
Daoud D, Mccarthy A, Dubetz C and Barker D (2018) The effects of emamectin benzoate or ivermectin spiked sediment on juvenile American lobsters (Homarus americanus). Ecotoxicology and Environmental Safety 163: 636–645. doi: 10.1016/j.ecoenv.2018.06.075
Duarte CM, Wu J, Xiao X, Bruhn A and Krause-Jensen D (2017) Can Seaweed Farming Play a Role in Climate Change Mitigation and Adaptation? Frontiers in Marine Science 4 doi: 10.3389/fmars.2017.00100
Edwards P, Zhang W, Belton B and Little DC (2019) Misunderstandings, myths and mantras in aquaculture: Its contribution to world food supplies has been systematically over reported. Marine Policy : 103547. doi: 10.1016/j.marpol.2019.103547
FAO (2018) Fishery and Aquaculture Statistics. Global capture production 1950-2016 (FishstatJ). In: FAO Fisheries and Aquaculture Department [online]. Rome. Updated 2018. FAO.
FAO (2020) The State of World Fisheries and Aquaculture 2020 – Sustainability in Action. Rome: FAO.
Fjørtoft HB, Nilsen F, Besnier F, et al. (2019) Salmon lice sampled from wild Atlantic salmon and sea trout throughout Norway display high frequencies of the genotype Associated with pyrethroid resistance. Aquaculture Environment Interactions 11: 459-468 doi: 10.3354/aei00322
Forney K, Southall B, Slooten E, et al. (2017) Nowhere to go: noise impact assessments for marine mammal populations with high site fidelity. Endangered Species Research 32: 391–413. doi: 10.3354/esr00820
Genschick S, Phillips MJ, Thilsted SH, Thorne-Lyman AL and Subasinghe R (2015) Aquaculture and fisheries for nutrition: towards a nutrition-sensitive approach. INFOFISH International 3: 11–17.
Golden CD, Seto KL, Dey MM, et al. (2017) Does Aquaculture Support the Needs of Nutritionally Vulnerable Nations? Frontiers in Marine Science 4 doi: 10.3389/fmars.2017.00159
Gorenzel WP, Conte FS and Salmon TP (1994) Bird Damage at Aquaculture Facilities. Prevention and Control of Wildlife Damage 15.
Hilborn R, Banobi J, Hall SJ, Pucylowski T and Walsworth TE (2018) The environmental cost of animal source foods. Frontiers in Ecology and the Environment 16: 329–335. doi: 10.1002/fee.1822
Jensen EM, Horsberg TE, Sevatdal S and Helgesen KO (2020) Trends in de-lousing of Norwegian farmed salmon from 2000–2019—Consumption of medicines, salmon louse resistance and non-medicinal control methods. PLOS ONE Public Library of Science.15: e0240894. doi: 10.1371/journal.pone.0240894
McSherry M, Davis RP, Andradi-Brown DA, et al. (2023) Integrated mangrove aquaculture: The sustainable choice for mangroves and aquaculture? Frontiers in Forests and Global Change Frontiers.6 doi: 10.3389/ffgc.2023.1094306
Marquez GPB, Santiañez WJE, Trono GC, et al. (2015) Chapter 16 – Seaweeds: a sustainable fuel source. In: Tiwari BK, Troy DJ, editors In: Seaweed Sustainability. San Diego: Academic Press.
Mongin M, Baird ME, Hadley S and Lenton A (2016) Optimising reef-scale CO 2 removal by seaweed to buffer ocean acidification. Environmental Research Letters 11: 034023. doi: 10.1088/1748-9326/11/3/034023
Nahuelhual L, Defeo O, Vergara X, et al. (2019) Is there a blue transition underway? Fish and Fisheries 20: 584–595. doi: 10.1111/faf.12354
Parker M and Bricker S (2020) Sustainable Oyster Aquaculture, Water Quality Improvement, and Ecosystem Service Value Potential in Maryland Chesapeake Bay. Journal of Shellfish Research National Shellfisheries Association.39: 269–281. doi: 10.2983/035.039.0208
Patricia Majluf et al. ,A review of the global use of fishmeal and fish oil and the Fish In:Fish Out metric.Sci. Adv.10,eadn5650(2024).DOI:10.1126/sciadv.adn5650
Pauly D (2019) Micronutrient richness of global fish catches. Nature 574 :41-42. doi: 10.1038/d41586-019-02810-2
Pechsiri JS, Thomas J-BE, Risén E, et al. (2016) Energy performance and greenhouse gas emissions of kelp cultivation for biogas and fertilizer recovery in Sweden. The Science of the Total Environment 573: 347–355. doi: 10.1016/j.scitotenv.2016.07.220
Quiñones RA, Fuentes M, Montes RM, Soto D and León‐Muñoz J (2019) Environmental issues in Chilean salmon farming: a review. Reviews in Aquaculture 11: 375–402. doi: 10.1111/raq.12337
Richardson, L. E., Lenfant, P., Clarke, L. J., Fontcuberta, A., Gudefin, A., Lecaillon, G., Le Vay, L., Radford, A. N., & Simpson, S. D. (2023). Examining current best-practices for the use of wild post-larvae capture, culture, and release for fisheries enhancement. Frontiers in Marine Science, 9, Article 1058497. https://doi.org/10.3389/fmars.2022.1058497
Shah S, Cabello F, L’Abée-Lund T, et al. (2014) Antimicrobial resistance and antimicrobial resistance genes in marine bacteria from salmon aquaculture and non-aquaculture sites. Environmental microbiology 16 doi: 10.1111/1462-2920.12421
Shepon A, Gephart JA, Henriksson PJG, et al. (2020) Reorientation of aquaculture production systems can reduce environmental impacts and improve nutrition security in Bangladesh. Nature Food Nature Publishing Group.1: 640–647. doi: 10.1038/s43016-020-00156-x
Taranger GL, Karlsen Ø, Bannister RJ, et al. (2015) Risk assessment of the environmental impact of Norwegian Atlantic salmon farming. ICES Journal of Marine Science 72: 997–1021. doi: 10.1093/icesjms/fsu132
Williamson T, Tilley D and Campbell E (2015) Emergy analysis to evaluate the sustainability of two oyster aquaculture systems in the Chesapeake Bay. Ecological Engineering 85: 103–120. doi: 10.1016/j.ecoleng.2015.09.052
Young M (2015) Marine animal entanglements in mussel aquaculture gear: Documented cases from mussel farming regions of the world including first-hand accounts from Iceland. University of Akureyri.
[1]This is calculated in terms edible weight; other statistics presented are calculated in terms of wet weight.



